 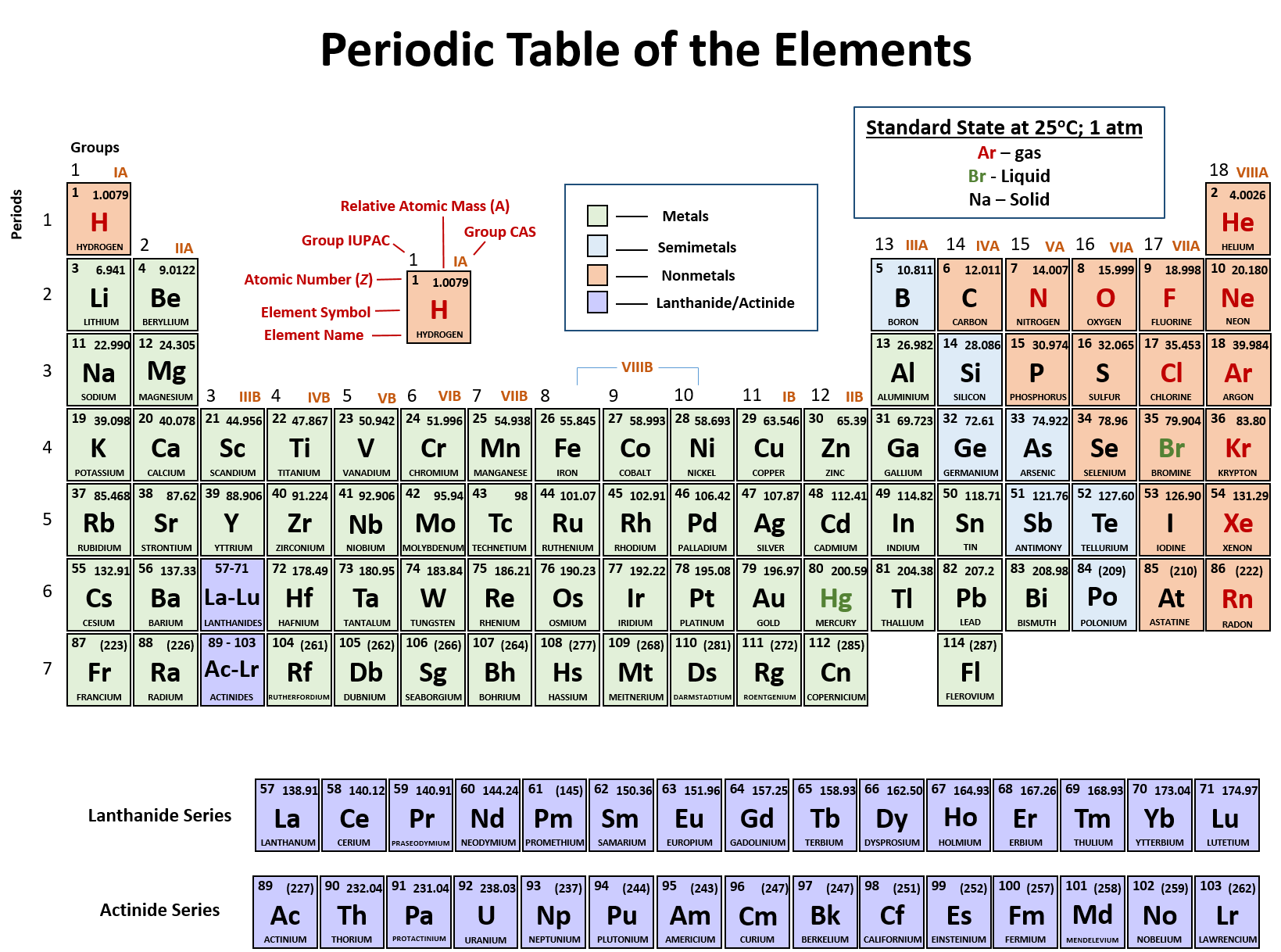
The first row is lanthanide series and the second row is actinide series. In addition to these seven rows, lanthanide and actinide series are shown separately at the bottom of the periodic table.Atomic numbers are serially indicated in the upper part of these boxes.The arrangement of the periods and groups results in the formation of boxes, where each box corresponds to the position of an element.The modern periodic table consists of seven horizontal rows called the periods and eighteen vertical columns called the groups.IUPAC recommends the term actinoid instead of actinide because the "ide" suffix is used for negatively charged ions.Īctinoid means "like actinium" so, strictly speaking, actinium should not be included in the list of actinoids, but it commonly is. You can find out more about lanthanoids and rare earth elements in the December 2015 issue of AUS-e-NEWS.ħ. Lanthanoid means "like lanthanum" so, strictly speaking, lanthanum should not be included in the list of lanthanoids, but it commonly is.įurthermore, the "rare earth elements" are made up of scandium, yttrium and the lanthanoid elements (including lanthanum). IUPAC recommends the term lanthanoid instead of lanthanide because the "ide" suffix is used for negatively charged ions. For High School students it is probably best to avoid the problems and use relative atomic mass (atomic weight) as a quantity with no units.Ħ. The issue will be further complicated if the dalton is redefined as being 0.001/N A kg since it would no longer be a unit of atomic mass relative to carbon-12. Since then the unit u (unified atomic mass unit) or Da (dalton) have been widely used but neither of these is an SI unit (as defined by the CIPM, International Committee for Weights and Measures). Prior to 1961 the unit amu (atomic mass unit) was used. A whole number is a counting or natural number, it does not include negative numbers, nor numbers which are fractions or decimals etc.ĥ. If you are interested in this topic, you can found out more in the September 2012 issue of AUS-e-NEWS.Ĥ. Once the element has been produced, it will be officially named and given an official symbol. These elements are assigned a name based on their atomic number which will have three numbers, so the symbol for these elements has three letters. The exception to this is for elements that have not yet been discovered. Don't worry about it, at High School you can treat all these terms as meaning the same thing.ģ. Debate still rages about whether the term atomic mass, atomic weight, relative atomic mass, relative atomic weight, or standard atomic weight should be used. The IUPAC periodic table uses standard atomic weights and provides the lower and upper bounds of the standard atomic weight for some elements. The element with atomic number 55 and symbol Cs may be spelt as caesium or as cesium.īUT it should be noted that these elements are present in the IUPAC periodic table as caesium and aluminium.Ģ. The element with atomic number 13 and symbol Al may be spelt as aluminium or as aluminum. The IUPAC recognizes the commonly used alternative spellings for two elements: Semi-metals lie between the metals on the left and the non-metals on the right of the periodic table.įootnotes: reference International Union of Pure and Applied Chemistry, IUPAC Periodic Table of the Elements, 28 th November 2016.ġ. Semi-metals or metalloids are written in orange text.Non-metallic elements are written in red text.Īll the non-metallic elements are found on the right hand side of the periodic table (except for hydrogen). 
Metallic elements are written in black text.The colour of the text in the boxes of the periodic table below shows the metallic character of the element: Gaseous elements are found in blue boxes.Liquid elements are found in lemon boxes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
